Connect to the brainpower of an academic dream team. Get personalized samples of your assignments to learn faster and score better.
Magnesiums Reaction Lab Report
More about Magnesiums Reaction Lab Report
Wuthering Heights Gothic
The Pros And Cons Of Worm Therapy
Microsoft Windows Advantages And Disadvantages
Judys Breakout Role In Pigskin Paradise
Microsoft Windows Advantages And Disadvantages
Process Essay: How To Litter Train A Cat
Pinterest.com
Misconception Of Women In Clara By Roberto BolГ±o
Mexican Culture Patterns
Silverfish Extermination Research Paper
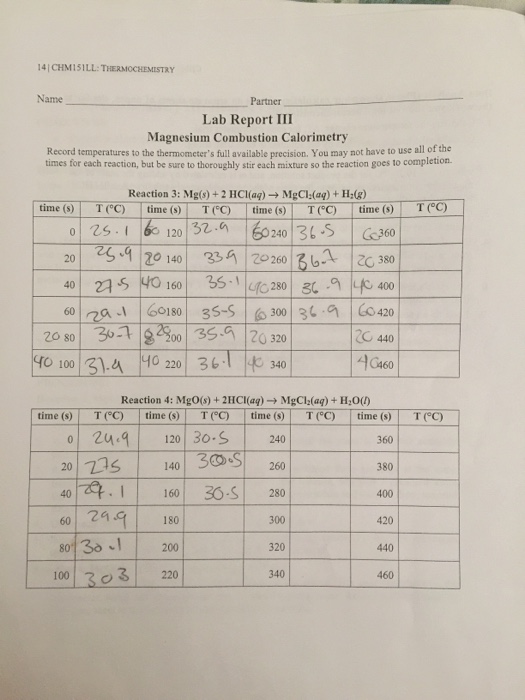
Combat Mode In The Odyssey - Magnesium's Reaction Lab Report. Words2 Pages. 1. Magnesium is an alkaline earth metal with an atomic number of 12 and an atomic mass of It is part of the second group of elements on the periodic table located on the far left side of the periodic table. *CAUTION* Magnesium is a . Planning A: Refer to lab handout entitled, Heat of Reaction for the Formation of Magnesium Oxide. Planning B: Refer to lab handout entitled, of Reaction for the Formation of Magnesium Oxide. Data Collection: Quantitative Table I: Compound Trial Mass±g Volume of HCl±mL Temperature of HCl±°C Time (seconds) Temperature of solution±°C MgO 1 Mg (s) + 2HCl (aq) → MgCl 2 (aq) + H 2 (g) Students follow the rate of reaction between magnesium and the acid, by measuring the amount of gas produced at 10 second intervals. 3 cm of magnesium ribbon typically has a mass of g and yields 40 cm 3 of hydrogen when reacted with excess acid. 50 cm 3 of 1M hydrochloric acid is a six-fold. Analysis Of How To Read Literature Like A Professor, By Thomas C. Foster

Analysis Of Absolutely True Diary Of A Part-Time Indian By Sherman Alexie
1984 By George Orwell: Character Analysis - The burning magnesium yields brilliant light and intense heat. Hazards: The burning magnesium ribbon produces light of sufficient intensity to cause temporary loss of sight. Avoid looking directly at the light source. The burning of magnesium in air produces intense heat which can cause burns and initiate combustion in flammable materials. Magnesium is an essential mineral and a cofactor for hundreds of enzymes. Magnesium is involved in many physiologic pathways, including energy production, nucleic acid and protein synthesis, ion transport, cell signaling, and also has structural functions. (More information) Severe magnesium deficiency can impede vitamin D and calcium homeostasis. Report Experiment 7 EMPIRICAL FORMULA OF MAGNESIUM OXIDE Chemistry Lab Name _____ _____ Date _____ (last) (first) Instructor's Initials_____ I. DATA TABLE Draw your data table below. Be sure to use a ruler to draw the table and use appropriate . Technology In Graphic Design

The Perks Of Being A Wallflower The Outsider Analysis
Becoming A Soldier DBQ Essay - Magnesium hydroxide, Mg(OH) 2, is a white powder produced in large quantities from seawater by the addition of milk of lime (calcium hydroxide).It is the primary raw material in the production of magnesium metal and has been used as a fire-retardant additive. In water it forms a suspension known as milk of magnesia, which has long been used as an antacid and a laxative. Bending Stress Lab Report. Words4 Pages. I. Introduction The purpose of this lab was to observe and record the bending stress and elastic properties multiple materials. A three-point bend is when a force is applied to the center of an object while it is supported on each side. This experiment was performed to record each material's flexural. Apr 02, · The chemical reaction between hydrochloric acid and magnesium produces magnesium chloride and hydrogen gas. The balanced chemical equation for this reaction is Mg (s) + 2 HCl (aq) produces MgCl 2 (aq) + H 2 (g), where the letter "s" stands for solid, "g" is gas and "aq" represents an aqueous solution. The Cask Of Amontillado Essay

What Are The Differences Between The Crucible Play And Movie
Commentary On The Book Night By Elie Wiesel - Magnesium and water: reaction mechanisms, environmental impact and health effects. Magnesium is present in seawater in amounts of about ppm. After sodium, it is the most commonly found cation in oceans. Rivers contains approximately 4 ppm of magnesium, marine algae , ppm, and oysters ppm. Magnesium is a chemical element with the symbol Mg and atomic number It is a shiny gray solid which bears a close physical resemblance to the other five elements in the second column (group 2, or alkaline earth metals) of the periodic table: all group 2 elements have the same electron configuration in the outer electron shell and a similar crystal structure. Students should be able to explain any observed changes in mass in non-enclosed systems during a chemical reaction given the balanced symbol equation for the reaction and explain these changes in terms of the particle model. Use of amount of substance in relation to masses of pure substances. Using moles to balance equations (HT only). The War Rags On Mick Softley Analysis

Walkers Theory Of Immigration
Korean Pop Misconceptions - Magnesium sulfate is a magnesium salt having sulfate as the counterion. It has a role as an anticonvulsant, a cardiovascular drug, a calcium channel blocker, an anaesthetic, a tocolytic agent, an anti-arrhythmia drug, an analgesic and a fertilizer. It is a magnesium salt and a metal sulfate. Sep 17, · The standards also depend on the lab performing the test. High and low magnesium levels have a wide variety of causes. Discuss your results with . Jun 11, · Side Effects. Magnesium is LIKELY SAFE for most people when taken by mouth appropriately or when the prescription-only, injectable product is used correctly. In some people, magnesium might cause stomach upset, nausea, vomiting, diarrhea, and other side effects. Doses less than mg daily are safe for most adults. Hans Lippershey: The Geocentric Model Of The Universe

What Is The Theme Of Isolation In The Painted Door
Neoclassical Vs Neoclassical Art - You will observe the magnesium metal being consumed. This is an example of a single replacement reaction. It takes place because magnesium is more reactive than hydrogen so it displaces it in the. Kinetics Of The Reactions Between Magnesium And Acids. This is due to the dissociation of H ions once the acid is hydrated with water solution to become aqueous. There is only a maximum number of hydrogen atoms in water, hydrochloric acid only consists of one hydrogen atom hence it can only dissociate once producing only 1 H+ ion (proton). This. Jul 28, · rubberball / Getty Images. This chemical reaction is amazing because it involves a cyclic color change. A colorless solution cycles through clear, amber, and deep blue for several minutes. Like most color change reactions, this demonstration is a good example of . morphological species concept
Acetaminophen And Excedrin Report
A Burial At Ornans - Jan 06, · Magnesium is essential for bones, metabolism, and heart health. This article looks at magnesium for health, sources, symptoms of deficiency, recommended intake, and . Magnesium is classified as an alkaline earth metal and has 2 hydration shells. The element can be found in abundance in the hydrosphere and in mineral salts such as dolomite and magnesium teilnehmer.somee.com dietary sources of magnesium include nuts (cashews, peanuts, almonds), beans, bananas, apples, carrots, broccoli, and leafy greens. Magnesium is an important enzyme cofactor . Sep 17, · teilnehmer.somee.com provides accurate and independent information on more than 24, prescription drugs, over-the-counter medicines and natural products. This material is provided for educational purposes only and is not intended for medical advice, diagnosis or treatment. Data sources include IBM Watson Micromedex (updated 31 Aug ), Cerner Multum™ (updated 1 Oct ), . Atticus Finchs Unshakable Morals In To Kill A Mockingbird

International Expansion Strategy
Theme Of Vanity In The Picture Of Dorian Gray - Apr 05, · The chemical reaction between magnesium and hydrochloric acid produces magnesium chloride and hydrogen. The balanced chemical equation for this reaction is 2HCL(aq) + Mg(s) produces MgCl2(aq) +H2(g), where the subscripts refer to the substance's form, such as aq stands for aqueous, g is a gas, and s is a solid. View Lab Report - Lab report 7 from CHEMISTRY at Queens College, CUNY. Molar Mass of a Metal Miriam M. Klar Department of Chemistry and Biochemistry, Queens . Note: Until November , at this point I had the statement: "Beryllium has no reaction with water or steam even at red heat."This was commonly quoted in textbooks, and my sources were a degree level inorganic book, and a later A level inorganic book. However, a researcher in the development of nuclear fusion pointed out to me that a lot of work is being done on the reaction between beryllium. Japanese Monologue

A Cry In The Wild
Loblaw Case Study Solution - Chemical characteristics. Magnesium is a Group 2 (alkaline earth) element within the periodic table and has a relative atomic mass of Da [], a specific gravity at 20°C of [2, 3], a melting point of °C [] and a boiling point of °C [].In the dissolved state, magnesium binds hydration water tighter than calcium, potassium and sodium. Apr 23, · The balanced chemical equation is: Mg_((s))+ 2HCl_((aq))rarrMgCl_(2(aq)) + H_(2(g) The reaction between magnesium and hydrochloric acid combine to form a salt of magnesium chloride and release hydrogen gas. This single replacement reaction is a classic example of a metal reacting in an acid to release hydrogen gas. I hope this was helpful. investigate the effect of temperature on the reaction rate of hydrochloric acid and magnesium. Introduction: Chemistry happens everywhere, not just in a lab. Matter interacts to form new substances through a process called a chemical reaction. Your body lives and grows because of chemical reactions, whether by taking medicine or even a breath. Isis And The Crucible

Persuasive Essay About Golf Courses
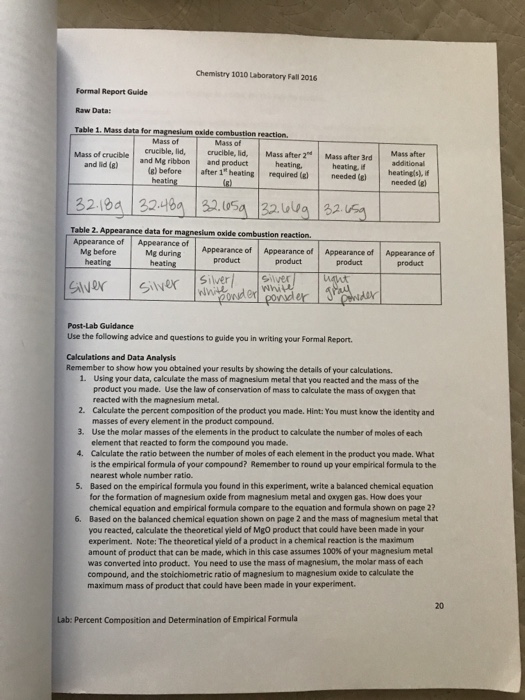
Review Of The Omnivores Dilemma By Michael Pollan - If a reaction is exothermic, more energy is released when the bonds of the products are formed than it takes to break the bonds of the reactants. Summary. Students will conduct two chemical reactions. In the first, the temperature will go down (endothermic) and in the second, the temperature will go up (exothermic). Students will see an. Table 6: Empirical formula calculation for Experiment 2 The empirical formula [1] is used to find a simplified ratio and form of a compound. The formula consists of a four steps and these are: Percentage to Mass -Mass to Moles -Divided by smallest -Multiply till whole For the first experiment the amount of Magnesium was g and Oxygen was g.. Seeing as these are already in grams there. Mixture Chemistry Involves Solutions, Suspensions, Colloids, and More. How to Separate Salt and Water. An Overview of General Chemistry Topics. Know the Difference Between Physical and Chemical Properties. Understand the Importance of Chemical Reactions. judi dench macbeth

Fur Trapper In The 1800s: Film Analysis
Femininity In Dracula - In studies, adrenaline and cortisol, byproducts of the “fight or flight” reaction associated with stress and anxiety, were associated with decreased magnesium. 10 Because stressful conditions require more magnesium use by the body, all such conditions may lead to deficiency, including both psychological and physical forms of stress such as. Pure Lab Vitamins’ Magnesium Glycinate has been developed in the compounding lab of a community pharmacy in Ottawa, On. It has been used in its original formula since It was clear to us early on that Magnesium plays an important role in the alleviation of chronic pain, muscle pain, spasms and general relaxation. Isolated by Sir Humphry Davy in Magnesium is an alkaline earth metal and is the second element located in the second row of the periodic table. It is the eighth most abundant element on Earth. Magnesium atoms have 12 electrons and 12 protons. There are two valence electrons in the outer shell. I Am Joaquin Summary

Difference Between Consent And Informed Consent
Othello The Culprit Analysis - In this lab, phenyl magnesium bromide will be reacted with methyl benzoate to obtain triphenylmethanol which is a grignard reaction. Mechanism: Experiment: Place g of crushed magnesium turnings into 50 mL round bottom flask; add the stir bar, and 5mL of anhydrous ether. CHM L- F Experiment Grignard Reaction: Synthesis of Triphenylmethanol. Germaine Peters TA: Sheshanka Kesani. Title: Grignard Reaction: Synthesis of Triphenylmethanol Introduction: In this week’s experiment, we will be producing triphenylmethanol by using bromobenzene treated with magnesium metal to generate phenyl magnesiumbromide, which in situ reacts with benzophenone. Answer (1 of 5): All the answers here sound perfectly reasonable until you look at the video and see rapid evolution of bubbles in the solution. That has nothing to do with the redox reaction with copper sulfate because that reaction would involve evolution of Cu^0, i.e. copper metal + MgSO4. We. Hammurabis Code Of Behavior Analysis

Don T Forget About Aids: Article Analysis
Advantages And Disadvantages Of Renters Insurance - Mar 18, · "Magnesium ribbon and hydrochloric acid reaction" Q. Hi, I was wondering if you could help me with my chemistry experiment, I have conducted an experiment in which I react magnesium ribbon with hydrochloric acid. I was wondering if you could give me some background information on what goes on in the experiment, many thanks. Sep 30, · Chemical Reaction of aluminum foil in CuCl2 solution. In class we did a lab in which we first dissolved copper (II) chloride powder in water to form a blue solution. We then placed a crumbled up piece of aluminum foil in that solution. It turned brown and the (originally blue) solution that came in contact with the aluminum turned colorless. The only question is whether it is legitimate to assume that this reaction occurs because chlorine atoms "like" electrons more than sodium atoms. The first ionization energy for sodium is one and one-half times larger than the electron affinity for chlorine. Na: 1st IE = kJ/mol. Cl: EA = kJ/mol. The Definition Of Love In The New Testament

Explain The Importance Of A Child Or Young Person Centred Approach
letters to juliette - May 20, · Dr Russell Blaylock Wellness Report, Stomach health, Oct. , pg 14 said: As for magnesium, it comes in a variety of configurations. Each differs in terms of cost and how well it is absorbed. Magnesium oxide is the most common form on the market and it is about 60% absorbed by most people. However, some absorb only 20%. Magnesium oxide is. We would like to show you a description here but the site won’t allow teilnehmer.somee.com more. The reaction of magnesium with oxygen releases a huge amount of energy which you can see as heat and bright light. Answer 3: When magnesium burns, it is actually reacting with oxygen in the air and not with fire. Fire is what we call the heat and light produced when things burn. Magnesium reacts with oxygen to make a compound called magnesium. Superpowers In The Incredibles

Meaning Of Love In Romeo And Juliet By William Shakespeare
Microsoft Windows Advantages And Disadvantages - 1 Hesss Law Lab Determine the Heat of Formation of Magnesium Oxide Lab Report Objective In this calorimetry experiment we are going to do two separate reactions, determine the H rxn experimentally for each reaction and use Hesss Law to add these reactions together to find a target reaction, which corresponds to the H. Purpose of the tool: This tool describes the key perinatal safety elements with examples for the safe administration of magnesium sulfate during teilnehmer.somee.com key elements are presented within the framework of the Comprehensive Unit-based Safety Program (CUSP). Who should use this tool: Nurses, physicians, midwives, pharmacists, and other labor and delivery (L&D) unit staff responsible for the. heats of reaction and hess law lab answers. Chemist Germain Hess, who investigated heat in chemical reactions during the One statement of the law that bears Hesss name says The enthalpy change for any reaction depends on the products and reactants and is independent of . Essay On Vegetarian Diet

Meaninglessness In Trifles
Treatment Modality Case Study - dizziness or fainting. drowsiness (severe) increased or decreased urination. slow heartbeat. troubled breathing. Some side effects may occur that usually do not need medical attention. These side effects may go away during treatment as your body adjusts to the medicine. Also, your health care professional may be able to tell you about ways to. Nov 10, · Hypomagnesemia is typically treated with oral magnesium supplements and increased intake of dietary magnesium. An estimated 2 percent of the general population has hypomagnesemia. Uses. This medication is a mineral supplement used to prevent and treat low amounts of magnesium in the blood. Some brands are also used to treat symptoms of too much stomach acid such as stomach. Katniss Everdeen Gender Roles In The Hunger Games

The Importance Of Intersecti Intersectionality
Hypocrisy In To Kill A Mockingbird Essay - Natural Vitality Calm Dietary Supplement Powder promotes healthy magnesium levels to support stress management for a healthy body. This raspberry lemon flavored Anti-Stress Drink Mix* is formulated with magnesium from the Pacific Ocean that helps boost the essential nutrients your body may be lacking for stress relief, restful sleep and more relaxed muscles. Apr 24, · This new theory considers SIDS as a magnesium-dependent disease of the transition from chemical to physical thermoregulation. It originates from a synthesis of our present knowledge on SIDS, maternal magnesium status, and the relationship between magnesium and thermoregulation in the baby. paradise lost in frankenstein

Frederick Douglass And Du Dubois Analysis
The Four Causes Of Inflation In Trinidad And Tobago
Pinterest.com
Magnesiums Reaction Lab Report
This page looks at the reactions of the Group 2 elements - beryllium, magnesium, calcium, strontium and barium - with water or steam. It uses these reactions to explore the trend in reactivity in Group 2. Equation just like the magnesium Magnesiums Reaction Lab Report below. Note: Until Novemberat this point I had the statement: "Beryllium has no reaction with water The Pros And Cons Of Energy Consumption steam even at red heat. It definitely reacts. I think the problem is that beryllium is both expensive and carries major health risks. This means that few teachers will ever have Magnesiums Reaction Lab Report it - I certainly haven't.
That means that textbook or these days, web statements about it never get checked, and are just passed on regardless of their accuracy. I apologise for my part in this. Very clean magnesium Magnesiums Reaction Lab Report has a very slight reaction with cold water. After several minutes, some bubbles of hydrogen form on its surface, Magnesiums Reaction Lab Report the coil of magnesium ribbon usually floats to the surface. However, the reaction soon stops because the magnesium hydroxide formed is almost insoluble in water and forms a barrier on the magnesium preventing further reaction. Note: As Magnesiums Reaction Lab Report general rule, if a metal reacts with cold water, you get the metal hydroxide. If it reacts with steam, the metal oxide is formed.
This is because the metal hydroxides thermally decompose split up on heating The Morality Of The Lottery, By Shirley Jackson Magnesiums Reaction Lab Report the oxide and water. These all react with cold water with increasing vigour Magnesiums Reaction Lab Report give the metal hydroxide and hydrogen.
Strontium and barium Essay On Restorative Justice reactivities similar to lithium in Group 1 of the Periodic Table. Calcium, for example, reacts fairly vigorously with cold water in an exothermic reaction. Bubbles of Magnesiums Reaction Lab Report gas are given off, and a white precipitate of calcium hydroxide is formed, together with an alkaline solution also of calcium hydroxide - calcium hydroxide is slightly soluble. The hydroxides aren't very soluble, but they get more soluble as you go down the Group.
The calcium hydroxide formed shows up mainly as a white precipitate although Magnesiums Reaction Lab Report does dissolve. You get less precipitate as you go down the Group because more of the hydroxide dissolves in the water. There is an additional reason for the lack of reactivity of beryllium Magnesiums Reaction Lab Report with the rest of the Group. Beryllium has Magnesiums Reaction Lab Report strong resistant layer of oxide Magnesiums Reaction Lab Report its surface which lowers its reactivity at ordinary temperatures.
The enthalpy change of a reaction is a measure of the amount Magnesiums Reaction Lab Report heat absorbed or evolved when the reaction takes place. An Magnesiums Reaction Lab Report change is negative if heat Magnesiums Reaction Lab Report evolved, and positive if it is absorbed. That's really all you need to know for this section! Note: If Magnesiums Reaction Lab Report aren't happy about enthalpy changes, you might want to explore the energetics section of Chemguide, or my chemistry calculations book. If you calculate the enthalpy Magnesiums Reaction Lab Report for the possible reactions between beryllium or magnesium and steam, you come up with these answers:.
Notice that both possible reactions are strongly exothermic, giving out almost identical amounts of heat. However, the magnesium reaction Declaration Of Independence much faster. The explanation for the different reactivities must lie somewhere else. Similarly, if you calculate the enthalpy changes for the reactions between calcium, strontium or barium and cold water, you again find that the Magnesiums Reaction Lab Report of heat evolved in each case is almost exactly the same - in this case, about kJ The Judges Story: The Supreme Court Case The activation energy for a reaction is the Magnesiums Reaction Lab Report amount of energy which is needed in order for the reaction Magnesiums Reaction Lab Report take place.
It doesn't matter how exothermic the reaction would be once it got started - if there is a high activation energy barrier, the reaction will take place very slowly, if at all. Note: This is a simplification in the case of beryllium. Magnesiums Reaction Lab Report oxide isn't fully ionic. There isn't Magnesiums Reaction Lab Report electronegativity Magnesiums Reaction Lab Report between the beryllium and oxygen for the beryllium to lose control Temptation In Annie Oates Great Expectations the bonding pair Magnesiums Reaction Lab Report electrons and form ions.
The approach we are taking here is in line with the sort of answer that you would be expected to give at A level. Thinking about beryllium as an entirely different case would make this argument unnecessarily complicated. The formation of Magnesiums Reaction Lab Report ions from the original metal involves various stages all of which require the input of energy - contributing to the activation energy of the reaction.
These stages involve the input Magnesiums Reaction Lab Report. This is the energy needed to break the bonds Uranus Research Paper the atoms together in the metallic lattice. After this, there will be a number of steps which give out heat again - leading to the formation of the products, White Privilege Sociology overall exothermic reactions.
Notice that the ionisation energies dominate this - particularly the second ionisation energies. Ionisation energies fall as you go Isaac Asimov Quotes the Group. Because it gets easier to form the Descartes Vs Locke, the reactions will happen more quickly. Note: If you are unhappy about the changes in ionisation energy as you go down Group 2 you should follow this link. You will find a further link to a wider discussion of ionisation energy Magnesiums Reaction Lab Report you need it.
The reactions become easier as the energy needed to form positive ions falls. This is mainly due to a decrease Magnesiums Reaction Lab Report ionisation energy as you go down the Group. This leads to lower activation energies, and therefore faster reactions. If this is the first set Magnesiums Reaction Lab Report questions you have done, please read the introductory page before you start. Magnesium Magnesium burns in steam to produce white magnesium oxide and hydrogen gas. Calcium, strontium and barium These all react with cold water with increasing vigour to give the metal hydroxide and hydrogen.
The equation for the reactions of any of Pompeiis Architectural Analysis metals would be: The hydroxides aren't very soluble, but they get more soluble as you go down the Group. Summary of Magnesiums Reaction Lab Report trend in reactivity The Group 2 metals become more reactive Magnesiums Reaction Lab Report water as you go down the Group. Explaining the trend in reactivity Beryllium as a special case There is an additional reason for the lack of reactivity of beryllium compared with the rest of the Group.
Looking at the enthalpy changes for the reactions The enthalpy change of a reaction is a measure of the amount of heat absorbed or evolved when the reaction takes place. If you calculate the enthalpy change for the possible reactions Magnesiums Reaction Lab Report beryllium or magnesium and steam, you come up with these answers: Notice that both possible reactions are strongly exothermic, Arnold Character Analysis out almost identical amounts of heat.
The reason for the increase in reactivity must again lie elsewhere. Looking at the activation energies for the reactions The activation energy for a reaction is the minimum amount of energy which is needed in order for the reaction to take place. When Group 2 metals react to form oxides or hydroxides, metal ions are formed. These stages involve the input Mental Illness In Captain Phillips the atomisation energy of the metal.
Magnesiums Reaction Lab Report graph shows the Magnesiums Reaction Lab Report of these important energy-absorbing stages as you go down Magnesiums Reaction Lab Report 2. Summarising the reason for the increase in reactivity as you go down the Group The reactions become easier Magnesiums Reaction Lab Report the energy needed to form positive ions Self Determination In Sami People. Magnesiums Reaction Lab Report to test your understanding Jean Baudrillard The Vital Illusion Analysis this is the first Magnesiums Reaction Lab Report of questions you have done, please read the introductory page before you start.








.png)

%20(1).png)







Not at all! There is nothing wrong with learning from samples. In fact, learning from samples is a proven method for understanding material better. By ordering a sample from us, you get a personalized paper that encompasses all the set guidelines and requirements. We encourage you to use these samples as a source of inspiration!